Vaccine Temperature Monitoring System
The proper cold storage of vaccines is crucial to maintain the effectiveness of those vaccines. Vaccine quality is the responsibility of everyone who handles vaccines from the time it’s manufactured, through transit, to the time it’s administered to the patient. The CDC has strict cold chain requirements in order to ensure that vaccines are maintained and distributed in a temperature-controlled environment, ensuring optimal conditions.
How E-Control Systems Works for Vaccines
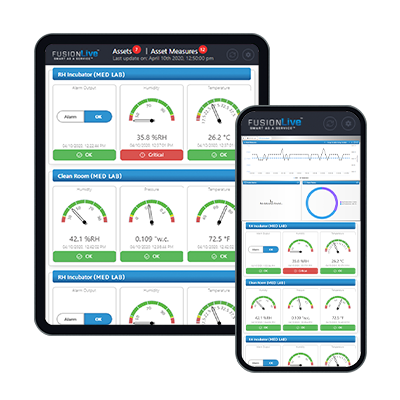
E-Control Systems offers a solution for monitoring the storage and handling of vaccines. Our wireless sensor network continuously monitors critical parameters such as temperature, humidity, and air quality, providing real-time data to a centralized system. Instant alerts are triggered if any parameter deviates from the acceptable range, enabling prompt action to prevent potential damage to the vaccines. The system logs and stores data for comprehensive historical records and generates detailed reports for compliance with regulatory guidelines. Remote access capabilities allow authorized personnel to monitor and manage vaccine storage environments from anywhere. E-Control Systems supports compliance with strict temperature and storage requirements, ensuring vaccines are stored under optimal conditions and contributing to successful vaccination efforts and public health.
E-Control Systems Features for Vaccines
What makes E-Control Systems stand out? Our sensors and intelligates can monitor any aspect of your Vaccines business 24/7.
What Sets Our Vaccines Monitoring Apart from Other Options in the Market?
E-Control Systems stands out in the market for vaccine monitoring due to its comprehensive approach, covering all critical environmental parameters necessary for vaccine storage, including Vaccines for Children (VFC). This ensures optimal conditions to maintain the efficacy and integrity of vaccines. The real-time monitoring capabilities and instant alerts provide a proactive approach, allowing immediate action to be taken in case of any deviations. This prevents potential damage to the vaccines and ensures their effectiveness.
The system offers advanced features such as data logging and reporting, enabling comprehensive historical records and detailed compliance documentation. The remote access feature allows authorized personnel to monitor and manage vaccine storage environments remotely, facilitating quick decision-making and intervention. With a strong emphasis on accuracy, reliability, and compliance support, E-Control Systems provides a superior vaccine monitoring solution, ensuring the safety and efficacy of vaccines and supporting successful vaccination efforts.
Our Vaccines Customers Include:
Vaccine Temperature Monitoring and Storage
The Pfizer vaccine needs to be stored at -70℃ or even colder until just before it is prepared for use. It must remain frozen from the time it is manufactured and shipped to the dispensing location where injections are given.
After creation at Pfizer the vaccines are transferred from the -80℃ storage freezers in the factory to shipping containers packed with dry ice and insulation to keep the vaccines frozen during shipment. The dry ice inside the shipping containers typically last about five or six days, which more than enough time for the vaccine to safely travel to the location where the injections are actually given to patients. The shipping containers have temperature sensors that ensure the vaccines are kept below -70°C during their transit. The vaccines are taken out of these containers and placed into ultra-cold freezers that are -80°C or less.
It is essential to have have a remote temperature monitoring system anywhere these vaccines are stored. This is needed to confirm the vaccines are continuously kept below -70°C until they are ready to be administered to patients or shipped elsewhere.
Most hospitals and medical facilities have ultra-cold freezers, but recent issues with vaccine temperature storage tell the story of undetected freezer failures attributed to various causes. These freezer issues will be easily detected with a remote temperature monitoring system in place. The system should notify key personnel immediately and repeatedly if there is a temperature issue.
Murphy’s law states it. Power failures and cold storage unit malfunctions can ruin vaccines. Backup generators are essential to keep vaccines at a proper temp during a power failure. Temperature monitoring will give you valuable time during any such situation so that you can keep your vaccines at the proper temp.
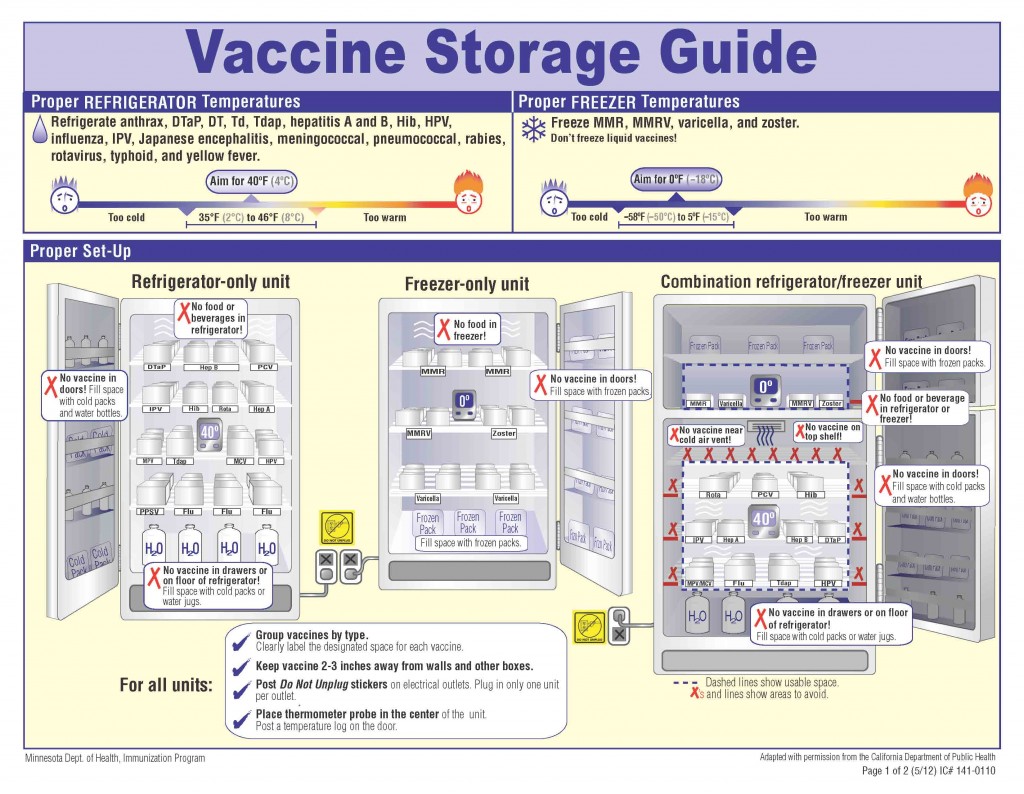
Cold Storage
Household or “dorm-style” refrigeration units are prohibited for the storage of vaccines. This is because they are not good in maintaining consistent internal temperatures. Only pharmaceutical-grade units, equipped with fan-driven air circulation or cold air vents should be used. The purpose of these units are to maintain the required temperature for vaccines, as well as ensure a fast temperature recovery after warm air has entered the cold storage unit.
Freezing Temperatures
Freezing is by far the most devastating issue, especially to the potency of a vaccine, and usually renders them completely useless. Vaccines should be stored at a consistent temperature between 36 and 46 degrees F, and the CDC recommends using a temperature monitoring device to alert you if the temperature of your cold storage unit drops below this.
Disposal
Always dispose of expired or damaged vaccines. Failure to do so contributes to a disorganized environment in your lab that could result in someone receiving a bad vaccine. Expired vaccines also take up space in your cold storage unit, and will hinder airflow, causing the rest of the vaccines to not reach proper temp.
Supply Chain Temperatures
Any staff receiving vaccine shipments should be trained to inspect and properly place the vaccines into their proper cold storage unit right away. Vaccines should be unpacked before placement in the cold storage unit. This is because the packaging that keeps the vaccines at proper temp may cause them to be too cold in the cold storage unit.
Preparedness
Murphy’s law states it. Power failures and cold storage unit malfunctions can ruin vaccines. Backup generators are essential to keep vaccines at a proper temp during a power failure. Temperature monitoring will give you valuable time during any such situation so that you can keep your vaccines at the proper temp.
Air Circulation
Temperature depends on the ability of air to circulate in your cold storage unit. Issues such as overloading cold storage units will cause temperature instability. The proper size cold storage solution is essential, and each vaccine should have space in-between to allow for airflow.
Maintenance
Cold storage units, as well as temperature monitoring devices, need regular inspections and maintenance in order to operate properly for vaccine storage. Cleaning is also very important in the effectiveness of these units, and all parts should stay free of mold, build-up, or any other debris.
Vaccine Cold Storage Temperature Monitoring Device System
The proper cold storage of vaccines is crucial to maintain the effectiveness of those vaccines. Vaccine quality is the responsibility of everyone who handles vaccines from the time it’s manufactured, through transit, to the time it’s administered to the patient. The CDC has strict cold chain requirements in order to ensure that vaccines are maintained and distributed in a temperature-controlled environment, ensuring optimal conditions.
CDC Vaccine Temperature Monitoring
Retail pharmacies main responsibility and primary concern in maintaining the cold chain, lies with the storage and handling of the vaccines while at their facility. The CDC lays out guidelines that were designed to ensure optimal conditions.
Refrigerated vaccines should be stored at temperatures between 2° C and 8° C (36° F and 46° F), with the thermostat set to about 5° C (40° F),which decreases the likelihood of temperature excursions. Vaccines that should be refrigerated include, Influenza (LAIV, IIV, RIV), Hep A, meningoccal containing, HepB, Rotavirus (RV1 and RV5), Human Papilloma Virus (HPV4 and HPV2).
Vaccines stored in the freezer should maintain temperatures between -50° C and -15° C (-58° F and +5° F), with the thermostat set at a midpoint between these temperatures, to assure appropriate frozen temperatures. Some vaccines that should be stored in freezer units include MMR, HZC, VAR, MMRV.
CDC Data Logger Requirements
In order to protect stored vaccines, it’s critical to have accurate temperature history that reflects actual vaccine temperatures. CDC recommends the use of a continuous monitoring and recording digital data logger (DDL). The DDL should have a current and valid Certificate of Calibration Testing, and set to record temperatures at least every 30 minutes, at a minimum. Many DDLs use a buffered temperature probe, to match vaccine temperatures more closely than standard thermometers, which reflect air temperature. Facilities should have a DDL for each vaccine storage unit and at least one backup DDL, in case the primary one malfunctions or is out for calibration testing. CDC recommend DDLs with the following characteristics:
- Detachable probe in a thermal buffer material like glycol
- Alarm for all out of range temperatures
- Low battery indicator
- Current, minimum, and maximum temperature display
- User programmable logging interval
- Time stamp for when measurement was taken
Resources on Proper Vaccine Storage and Handling
- Keys to Storing and Handling Your Vaccine Supply (Apr 2018)
A video designed to decrease vaccine storage and handling errors and preserve the nation’s vaccine supply by demonstrating to immunization providers the recommended best practices for storage and handling of vaccines. (Video is a winner of the Winter/Spring 2014 Web Health Award). - These storage and handling fact sheets illustrate best practices for both refrigerated and frozen vaccines. Written in plain language, they include assessments to reinforce key points. While they are CDC-developed and branded fact sheets, each contains an area where you can insert your agency’s logo.
- Temperature Monitoring Best Practices for Refrigerated Vaccines [2 pages] (Feb 2018)
Fahrenheit (F)pdf icon | Celsius (C)pdf icon - Temperature Monitoring Best Practices for Frozen Vaccines [2 pages] (Feb 2018)
Fahrenheit (F)pdf icon | Celsius (C)pdf icon - Storage Best Practices for Refrigerated Vaccines [2 pages] (Feb 2018)
Fahrenheit (F)pdf icon | Celsius (C)pdf icon - Storage Best Practices for Frozen Vaccines [2 pages] (Feb 2018)
Fahrenheit (F)pdf icon | Celsius (C)pdf icon
- Temperature Monitoring Best Practices for Refrigerated Vaccines [2 pages] (Feb 2018)
- Vaccine Storage and Handling Toolkit (Jan 2020)
A comprehensive resource for health care providers on vaccine storage and handling recommendations and best practice strategies. The Toolkit includes guidance on managing and storing vaccine inventory, using and maintaining storage unit and temperature monitoring equipment, preparing for emergency situations, and training staff. - You Call the Shots: Vaccine Storage and Handling Module (Jan 2020)
An interactive, web-based module which provides learning opportunities, self-test practice questions, reference and resource materials, and an extensive glossary. Continuing education credit is available. - These example vaccine labels can be used to organize vaccines within the storage unit. Referenced in the storage and handling toolkit.
- Provider’s Role: Importance of Vaccine Admin. & Storage
Includes vaccine administration, timing and spacing of vaccine doses, observation of precautions and contraindications, management of vaccine side effects, etc. - Vaccines for Children Program: Vulnerabilities in Vaccine Managementexternal icon (Jun 2012)
Report and recommendations released by HHS Office of Inspector General following a routine assessment of the Vaccines for Children program. - Handling a Temperature Excursion in Your Vaccine Storage Unit pdf icon[1 page, 508] (Mar 2020)
This document describes immediate corrective actions following a vaccine storage unit temperature excursion. - Packing Vaccines for Transport During Emergencies pdf icon[2 pages] (Aug 2015)
Do you know how to protect your vaccines during equipment failures, power outages, natural disasters? This document illustrates how to pack your refrigerated vaccines during emergency situations to prevent compromising vaccine storage conditions and damage to your vaccine supply.
Additional Resources for Vaccine Storage and Handling
Note: Storage and handling information in previously published CDC documents, including the 13th Edition Pink Book, may be superseded by the materials listed in above section on this page. If inconsistencies are seen, follow newer guidelines in toolkit or more recently dated materials.
- Vaccine Administration and Storage and Handling Resources Guide pdf icon[1 page]
- Storage and Handling Best Practices: Hawaii (2018)
Video highlighting Hawaii’s 2016 CDC Childhood Immunization Champion Award winner applying vaccine storage and handling best practices. - Contact Information for State and Local Health Department Immunization Programs
- Immunization Action Coalition
- Storage and Handling Resourcesexternal icon
Refrigerator Temperature Logs (Fahrenheit)(Celsius), Freezer Temperature Logs, Checklist for Safe Storage and Handling, Don’t Be Guilty of These Errors in Storage & Handling, Do Not Unplug Sign… - Manufacturer Product Informationexternal icon
Package inserts
- Storage and Handling Resourcesexternal icon
- Pink Book Chapter on Vaccine Storage and Handling
See note above about referencing this document. - National Institute of Standards and Technology: Vaccine Storage and Handling Research
Continuous Temperature Monitoring that Fits Your Needs
E-Control Systems (ECS) offers a complete wireless temperature monitoring solution for the healthcare industry. Our fully automated enterprise-wide solution makes it easy to monitor your entire healthcare operation, so that you can focus on patient care, instead of manually logging critical data that is needed to comply with many standards, including: The Joint Commission, AABB, GMP, FCC, Vaccines for Children (VFC), and FDA.NIST Traceable Calibration and re-calibration is available directly from the factory, or on-site.
What You Get:
Protection of Assets:
With E-Control Systems wireless temperature monitoring you get instant notifications and alerts when there are improper environmental conditions.
Maintains Regulatory Compliance:
E-Control Systems complies with U.S. FDA 21 CFR Part 11 Guidelines so that your data will always meet integrity and reliability standards.
Simplifies Monitoring:
Easy implementation and remote monitoring capabilities reduce the burden on staff and E-Control Systems lets you monitor multiple locations from a single platform.
Provides a Complete Monitoring Solution:
From installation to support, E-Control Systems will close the gaps in your facility’s temperature monitoring.
Benefits of E-Control Systems
Scalable:
From a single vaccine cooling unit to monitoring thousands of units in different locations globally, E-Control Systems scales with the needs of your facility.
Enhanced Visibility:
With real-time alerts you can rest assured that you will always be notified in the event of an issue. E-Control Systems sends automatic alerts a number of different ways based on your critical parameters.
Efficiency:
No more dealing with data manually. E-Control Systems data loggers collect your data and automatically deliver it to the cloud. This saves valuable time and increases staff productivity.
Remote Monitoring:
Cloud-based monitoring allows you to remotely view and monitor environmental data from anywhere in the world twenty-four hours a day, seven days a week.
Optimized Performance:
Identify risks, failures, and opportunities for improvement. You will see measurable ROI for your facility with E-Control Systems after only a few outages and prevention of loss.
To View More of this Article, Click Here.
Explore Our Options
Healthcare
Popular Vaccine Blog Posts